-
Photothermal therapy has emerged as a promising modality in interventional oncology, enabling localised energy deposition using minimally invasive probes1,2. Optical fibres, in particular, provide a low-loss and geometrically compliant pathway for light delivery to deep-seated tumours and have gained growing interest as versatile platforms for interventional applications3. Recent advances have demonstrated fibre-based systems with expanded therapeutic and diagnostic capabilities that integrate localised photothermal treatment, drug delivery, and in situ optical readouts in compact fibre geometries4–6. However, most existing fibre-based approaches are limited to single-function implementations within individual fibres, whereas attempts to integrate multiple functions are frequently hindered by interfunctional crosstalk caused by spectral overlap among the optical reagents involved7–9. These constraints have led to the prevalent adoption of multifibre or multicomponent configurations at the expense of increased bulk and stiffness, which may compromise interventional compatibility and tissue safety. Spectrally multiplexed strategies combined with fluorescent functional materials have emerged as promising alternatives10,11, enabling compact multifunctional integration within a single fibre. By assigning distinct excitation and emission wavelengths to different functions, spectral multiplexing provides an intrinsic mechanism for decoupling the sensing, therapy, and readout channels within the same fibre, thereby suppressing interfunctional crosstalk without increasing structural complexity.
In a recently published paper on Light: Science and Applications, Li et al. presented a single-fibre integration strategy that consolidates tumour therapy and multimodal sensing within a unified fibre architecture (Fig. 1)12. Spectrally addressable functional components were co-embedded in a hydrogel matrix at the distal end of a tapered optical fibre, enabling pH and temperature sensing, along with photothermal therapy at the fibre tip. The local pH was monitored using a fluorescent pH indicator (HPTS-IP, derivative of 8-hydroxy-1,3,6-pyrene trisulfonic acid), which was excited at 405 and 450 nm with a ratiometric emission centred at approximately 520 nm. This dual-excitation ratiometric scheme enables a robust pH readout that is insensitive to HPTS-IP concentration and optical coupling variations during in situ measurements. Temperature sensing was achieved using a lanthanide-based metal-organic framework (Ln-MOF) probe whose emission exhibits temperature-dependent behaviour. The Ln-MOF was excited at 295 nm in the ultraviolet, producing characteristic emissions at 564 and 618 nm, and the relative intensity of these bands provided a quantitative measure of the local temperature. Notably, the excitation and emission bands of the temperature probe were isolated spectrally from those of the pH sensor. To generate localised heating for tumour ablation, indocyanine green (ICG) is employed as a photothermal agent and excited at 808 nm. This near-infrared excitation band is spectrally well-separated from the fluorescence excitation and emission windows used for sensing, thereby minimising interference between the therapeutic and sensing channels.
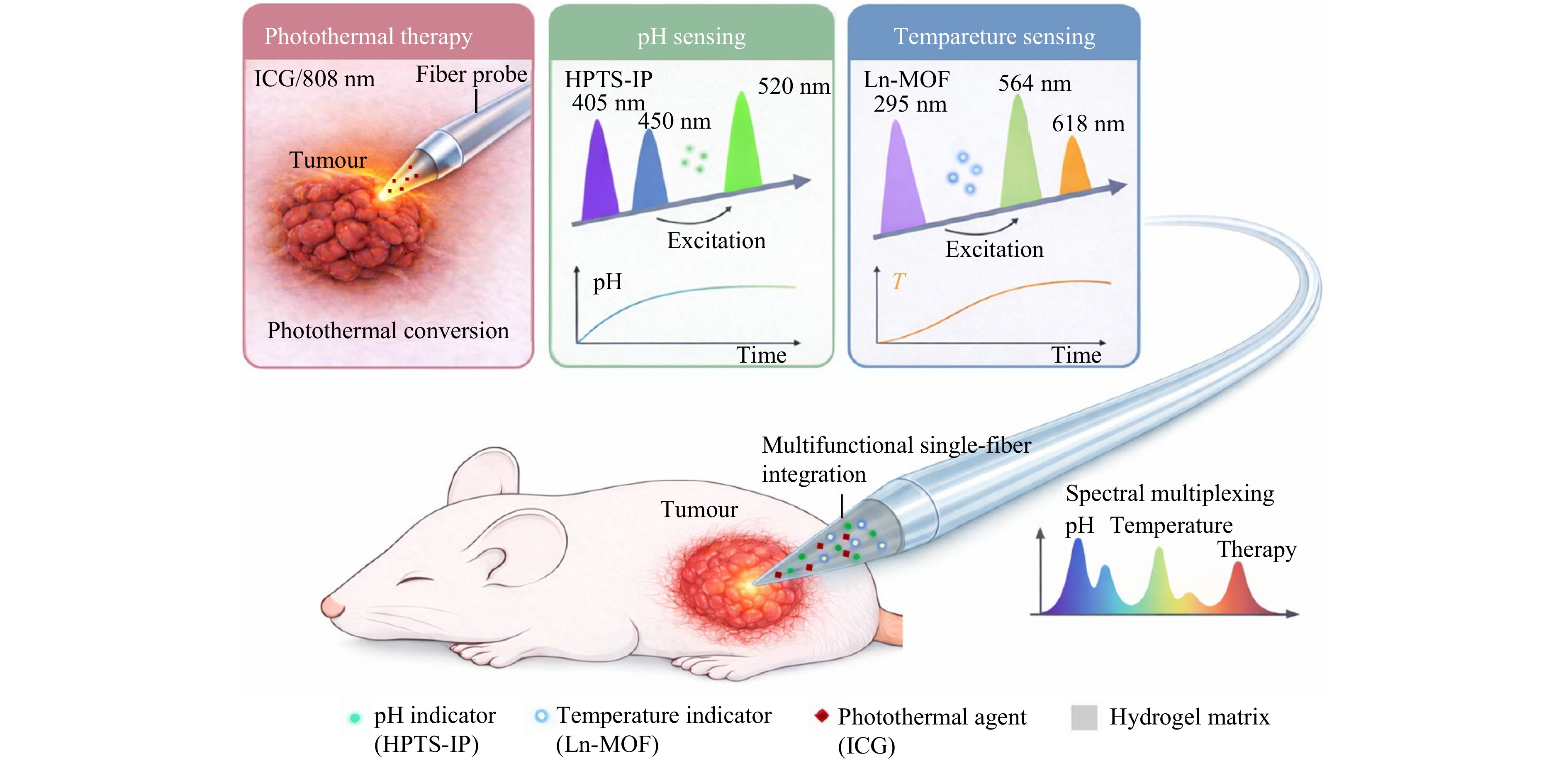
Fig. 1 Closed-loop tumour photothermal therapy enabled by spectrally multiplexed multifunctional single-fibre integration.
Using this single-fibre platform, they further demonstrated closed-loop tumour photothermal therapy in vivo across the pre-, intra-, and post-treatment stages in a subcutaneous colorectal cancer xenograft mouse model. Prior to treatment, the probe utilised spatial variations in the local pH to identify the tumour boundaries. Following insertion into the tumour tissue and adjacent normal regions, fluorescence-based pH readouts revealed a characteristic pH gradient between malignant and healthy tissues, enabling the delineation of tumour edges and informed positioning of the fibre for precision therapy. During treatment, the photothermal channel was activated, and the embedded photothermal agent converted the guided laser energy into localised heat for tumour ablation. Simultaneously, real-time temperature readouts acquired at the fibre tip provided continuous feedback on the local thermal conditions, allowing the adjustment and maintenance of photothermal dosing within a controlled range. After treatment, real-time monitoring of the local pH can be used to rapidly assess therapeutic efficacy, as the reversal of tumour acidity reflects the microenvironmental improvement induced by tumour cell damage. An increase in local tumour pH indicates the alleviation of the acidic tumour microenvironment and serves as an immediate functional indicator of treatment response. The sequential use of pH sensing for tumour localisation, temperature feedback for intratreatment control, and pH monitoring for posttreatment evaluation establishes a closed-loop operational paradigm in which diagnosis, therapy, and outcome assessment are temporally integrated, advancing more precise and adaptive interventional therapies.
The significance of this study lies in redefining the role of a single optical fibre in interventional therapy. Such a closed-loop fibre platform is not intended to operate in isolation but is inherently compatible with existing interventional frameworks. Well-established fibre-based imaging technologies, including optical coherence tomography probes and catheter-based photoacoustic systems, have already demonstrated how high-resolution structural and functional information can be acquired through ultrathin optical fibres during intervention13–16. Integrating these imaging capabilities with the feedback-guided therapeutic fibre demonstrated here could further enrich local situational awareness during treatment. Recent advances in fibre-enabled and soft robotic systems for minimally invasive interventions highlight how navigation, actuation, and sensing can be combined in compliant architectures17,18. Against this backdrop, the present study provides a missing therapeutic counterpart, paving the way for the redefinition of fibre-centric interventional systems in which imaging, navigation, and feedback-guided energy delivery are coherently integrated within established clinical workflows.
Multifunctional single-fiber integration enables closed-loop tumour photothermal therapy
- Light: Advanced Manufacturing , Article number: 55 (2026)
- Received: 25 February 2026
- Revised: 17 March 2026
- Accepted: 18 March 2026 Published online: 29 April 2026
doi: https://doi.org/10.37188/lam.2026.055
Abstract: Interventional photothermal therapy in oncology requires accurate tumour targeting, controlled thermal dosing, and timely feedback. Optical fibres offer a compact route for light delivery; however, most fibre-based approaches remain functionally fragmented. A recent study reports a multifunctional single-fibre probe that integrates tumour identification, photothermal therapy, and real-time feedback through wavelength-division strategies, representing an important step toward closed-loop tumour photothermal therapy.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article′s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article′s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.





 DownLoad:
DownLoad: